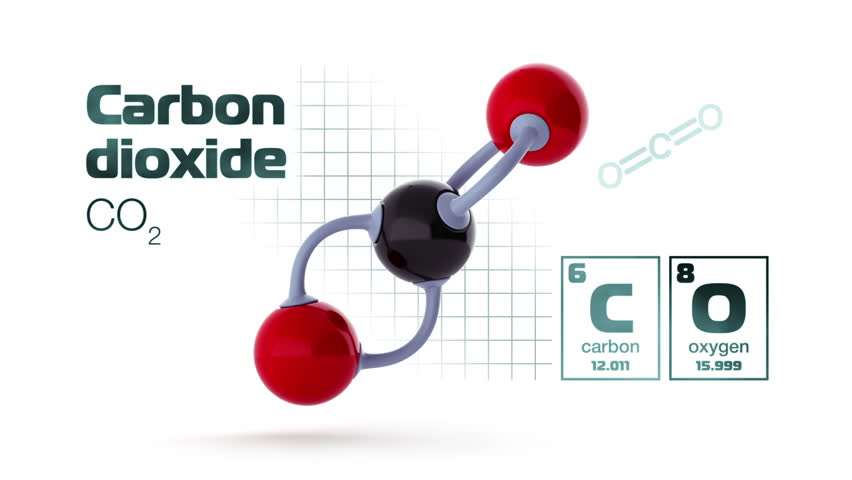
If you start with 100 g of hydrogen that reacts in this entire process in an excess of carbon dioxide and oxygen … What is the balanced equation when barium reacts with water. The balanced chemical equation for C3H8 O2 = CO2 H2O is C3H8 5O2 = 3CO2 4H2O. c) Aluminum copper chloride → Aluminum chloride Copper. How do you write a balanced chemical reaction for this process? Chemistry. When dry, baking soda doesn't decompose very quickly, although it does have a shelf life, so you should test it before using it as a cooking ingredient or in an experiment. The Balanced Equation For This Reaction Is: 2 C4H10 (g) 13 O2 (g) 8 CO2 (g) 10 H2O (g) If 8 Moles Of Butane (C4H10) React, The Reaction Consumes ? The oxide forms of carbon you mention are oxidation products of carbon. Find a balanced chemical equation for this reaction. Oxygen thus acts as the oxidant and removes electrons from the carbon. carbon dioxide water → glucose oxygen. How are the 3 mutated strands similar to each other? On the right side there are: 1 calcium atom. Carbon and oxygen are the reactants, and carbon dioxide is the product. 
Explanation Explanation Check Check So we put a 4 in front of the CO on the RHS to give: CO_2 O_2 \Rightarrow 4CO. What is the balanced equation for the chemical reaction #Mg_3N_2 2H_2O -> Mg(OH)_2 NH_3#? 3 oxygen atoms. Write a balanced equation showing all reactants and all reaction products in the proper proportions. Agent Ruganiego otwiera temat odejścia swojego klienta The balanced chemical reaction is Ca(OH)2 CO2 -> CaCO3 H2O. Chemistry Chemical Reactions Balancing Chemical Equations. Salvini: Gra AC Milanu to nie tylko wina Gattuso. What Is The Balanced Equation Of Photosynthesis? 4Fe(s) 3O2(grams) => 2Fe2O3(s) chemistry This formula could also be read as: Glucose oxygen –> water carbon dioxide energy. The balanced equation for the reaction is: The decomposition of anhydrous sodium carbonate into sodium oxide and carbon dioxide occurs slowly at room temperature and proceeds to completion at 851 C (1124 K). The combustion of butane in oxygen produces carbon dioxide and water. iron (III) oxide is formed when iron combines with oxygen in the air. d) Hydrogen Chlorine → Hydrogen chloride. how many grams of Fe2O3 are formed when 16.7g of Fe reacts completely with oxygen? Write A Balanced Chemical Equation For This Reaction. The equation for the reaction of carbon and oxygen to form carbon dioxide is deceptively easy to write. 
it contains 70.5% carbon, 11.5% hydrogen, and 10.4% oxygen by mass. One particular soap molecule has 18 carbon atoms. Carbon monoxide (g) oxygen(g) -> carbon dioxide(g).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
