|
Yet, recent experiments revealed the existence of two adders, one on cell division and the other on replication initiation, and that these two processes are more loosely coupled than hitherto believed 8, 18, 19, 20, 21, 22, 23. It has been proposed that cell division control is tightly coupled to the control over replication initiation 3, 16, 17, via a sizer on replication initiation and a timer for cell division. These experiments revealed that cells obey an adder principle, which states that cells add an on average constant volume independent of the birth volume during each cell cycle. To obtain insight into the mechanisms that control DNA replication and cell division, fluctuations in cell size have been studied 14, 15. In the LDDR model, replication forks overlap and RIDA is the main deactivator in combination with the activators DARS1 and DARS2. d Scheme of the initiator switch models: In the LD model, ATP-DnaA is mainly activated via the acidic phospholipids and deactivated via the site datA. coli, the initiator DnaA (red circles) is negatively autoregulated with the dissociation constant \(\), respectively. DnaA also has a high affinity for titration sites (gray circles) located on the DNA. DnaA is activated via the acidic phospholipids in the cell membrane and via the two chromosomal sites DARS1 and DARS2, and deactivated via the chromosomal site datA and via regulatory inactivation of DnaA (RIDA), a process coupled to active DNA replication. Replication is initiated by the ATP-bound form of the initiator protein DnaA. coli chromosome: Replication starts at the origin ( oriC, yellow circle) and proceeds via two replication forks to the terminus (ter, gray bar). Initiating replication at a constant origin density ρ * (dashed red line) or respectively a constant volume per origin v * = 1/ ρ * (dashed green line) and division a constant time τ cc later (blue arrows) ensures that the cell initiates replication once per division cycle and that it maintains cell size homeostasis at slow (light blue regime) and fast (dark blue regime) growth rates. 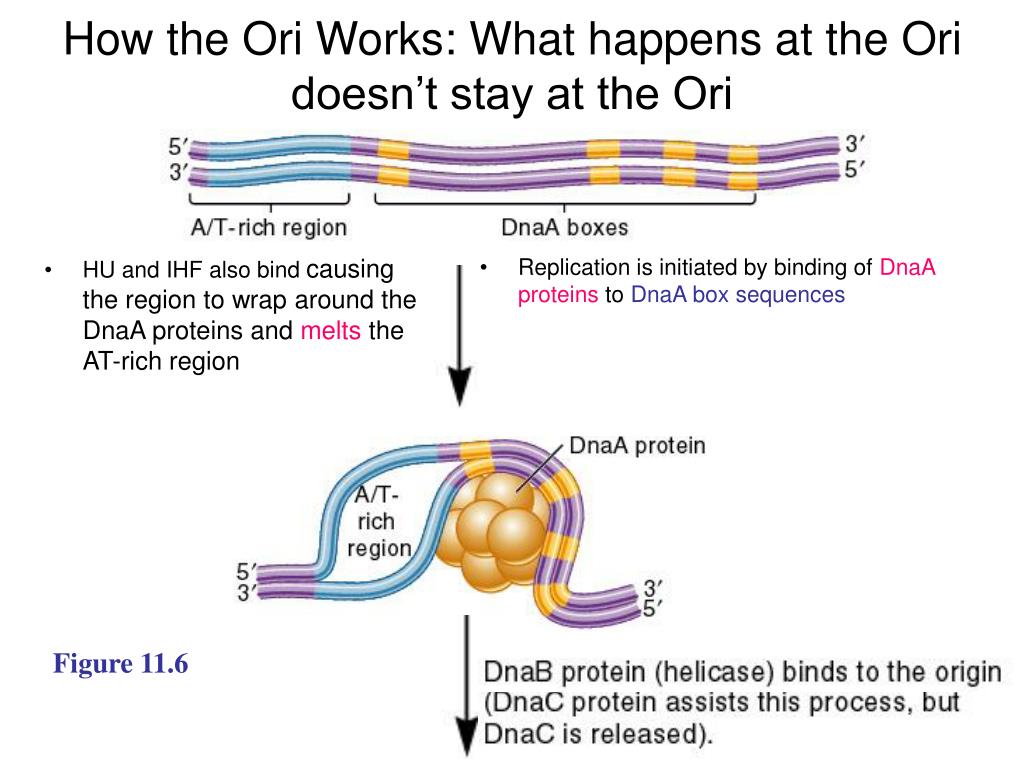
Yet, how the initiation volume is controlled so precisely, and what molecular mechanism gives rise to robust cell cycles over many generations remains despite extensive studies poorly understood 9, 10, 11, 12, 13.Ī The volume V( t), the number of origins n ori( t) and the origin density ρ ori( t) = n ori( t)/ V( t) as a function of time. Moreover, single-cell measurements revealed that the initiation volume is one of the most tightly controlled cell-cycle parameters, varying by about 10% for any measured growth rate 3, 8. Recent experiments at the population level showed that the average initiation volume per origin v * varies within a ~50% range over a tenfold change in the growth rate 7. Initiating replication at a constant origin density ensures that DNA replication is initiated once per cell cycle per origin, which is a necessary condition for maintaining stable cell cycles at all growth rates (Fig. Donachie then predicted that replication is initiated at a constant volume per origin v * 6. This apparent paradox was resolved by the model of Cooper and Helmstetter in which new rounds of replication are initiated before the previous round has finished 5 (Fig. Intriguingly, in nutrient-rich environments, the model organism Escherichia coli can even divide faster than the time it takes to replicate its entire chromosome 1, 2, 3, 4. To maintain stable cell cycles over many generations, living cells must coordinate DNA replication with cell growth and cell division. In addition, it reveals how an origin-density sensor yields adder correlations. Our analysis thus predicts that both mechanisms together drive robust replication cycles at all growth rates. Conversely, while the activation switch alone yields robust rhythms at high growth rates, titration can strongly enhance the stability of the switch at low growth rates. 
In this regime, the DnaA activation switch becomes essential for stable replication initiation. We first show that a mechanism solely based on titration generates stable replication cycles at low growth rates, but inevitably causes premature reinitiation events at higher growth rates. Here, we study by mathematical modelling how these two mechanisms interact to generate robust replication-initiation cycles. 
coli controls replication initiation via titration and activation of the initiator protein DnaA. Yet, a molecular model that can explain these observations has been lacking. The bacterium Escherichia coli initiates replication once per cell cycle at a precise volume per origin and adds an on average constant volume between successive initiation events, independent of the initiation size.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
